Continuing my recent history of only brief notes (sorry, busy…) here a short headline from the SPIE Optics and Photonics Conference in San Diego.
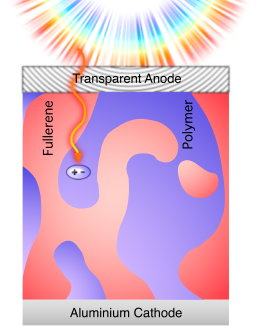
Today I heard a talk by Darin Laird, Plextronics.  Using an undisclosed organic donor material (well, they call their product Plexcore OS 2000 [Update below], as opposed to their P3HT OS 1000 or so) blended with the usual suspect PCBM, they managed to process an NREL certified lab scale (0.1cm2) solar cell with 5.94% power conversion efficiency! Fill factor was almost 72%, I believe, with the major improvement as compared to the reference material P3HT coming from an increased open-circuit voltage.
Using an undisclosed organic donor material (well, they call their product Plexcore OS 2000 [Update below], as opposed to their P3HT OS 1000 or so) blended with the usual suspect PCBM, they managed to process an NREL certified lab scale (0.1cm2) solar cell with 5.94% power conversion efficiency! Fill factor was almost 72%, I believe, with the major improvement as compared to the reference material P3HT coming from an increased open-circuit voltage.
The corresponding solar cell module, 15×15 cm2 large, has an efficiency of 1.1% (or 2.3% active area efficiency, if you consider that only 46% of the module are active area). These numbers are brand new, but generally, uptodate solar cell efficiencies can be found in the efficiency tables (V32) by Martin Green.
So, who’s next to boost the organic solar cell efficiencies? ;-)
P.S. As there sadly was a history of overestimated efficiencies published, followed by letters to the editors by watchful scientists and statements, a solar cell characterised by a certified institute is important to regain the trust.
P.P.S. Of course, not every university group can afford to spend 1000 bucks on a certified solar cell measurement. Still, at least some effort can be put into doing the current-voltage characterisations carefully. In January, Jan Kroon gave an interesting talk about measuring organic solar cells properly; find the video here.
Update (5.9.2008): The donor Plexcore OS 2100 available at Sigma Aldrich is not the one with which the 5.9% efficiency where achieved. The undisclosed donor material used is not yet available commercially, it seems.



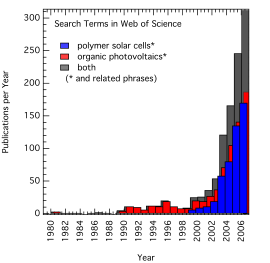 I just updated it using
I just updated it using 


