It seems that one prominent discussion in organic photovoltaics has officially ended, the one about the primary photoexcitation in disordered organic solar cells being excitons (with a binding energy clearly above 100meV) or free charges (with excitons having binding energies in the range of the thermal energy, i.e. <<100meV). Hwang, Moses and Heeger, have just published a paper on polymer:fullerene blends [Hwang 2008] where they describe the charge generation as
Mobile carriers are generated via a two-step process: initial ultrafast charge separation to an intermediate charge transfer (CT) bound state, followed by the transfer of carriers onto the bicontinuous networks.
They explicitly mention
[…] indicating ultrafast dissociation of the singlet excitons at the polymer-PCBM interface and the build-up of the initial CT state.
The paper is nice but in itself not that remarkable, except that previously, Moses and Heeger always claimed the primary photoexcitation to be free charges instead of bound excitons. Their measurements yielded exciton binding energies in the range of the thermal energy, i.e., no donor acceptor interface being necessary for charge separation. To quote an older paper [Moses 2000],
Thus, carriers are photoexcited directly and not generated via a secondary process from exciton annihilation.
Now I have to mention that in the new paper they use P3HT:PCBM, and in the old one MEH-PPV:PCBM. But as they do not mention this in the new paper, I assume that either I missed something, or they changed their point of view concerning the primary photoexcitation.

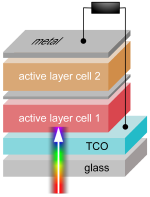
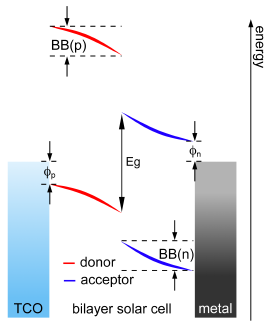


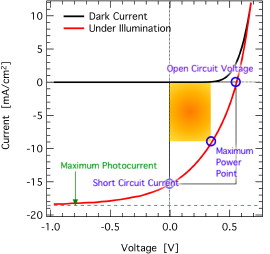 made for crystalline inorganic diodes, when applied on fitting organic solar cells.
made for crystalline inorganic diodes, when applied on fitting organic solar cells.

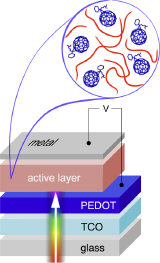 The vision for the so called plastic electronics is to print circuits and devices on flexible substrates. This can be done at room temperature (low energy) and ideally with roll-to-roll processes (high throughput). Sounds good, eh?
The vision for the so called plastic electronics is to print circuits and devices on flexible substrates. This can be done at room temperature (low energy) and ideally with roll-to-roll processes (high throughput). Sounds good, eh?