In order to improve the power conversion efficiency of organic solar cells, novel donor and acceptor materials will have to be synthesised. 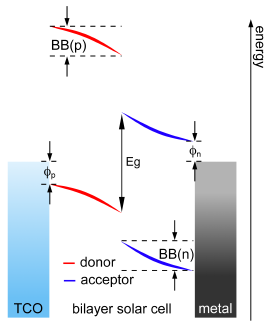 Properties looked for are the ability to self-organise – enhancing order and thus charge transport – and an absorption spectrum as wide as possible, being one of the major limiting factors as of yet. Nowadays, in most cases only the donor material absorbs light efficiently; an absorbing acceptor has a large potential for increasing the photocurrent. Additionally, by a variation of the relative energy levels of donor and acceptor material, the energy loss due to the electron transfer can be minimised: For light absorption in the donor, it is hoped that if the energy offset between donor LUMO (lowest unoccupied molecular orbital) and acceptor LUMO is a tiny bit larger than the exciton binding energy, a positive impact on the open-circuit voltage will be seen.
Properties looked for are the ability to self-organise – enhancing order and thus charge transport – and an absorption spectrum as wide as possible, being one of the major limiting factors as of yet. Nowadays, in most cases only the donor material absorbs light efficiently; an absorbing acceptor has a large potential for increasing the photocurrent. Additionally, by a variation of the relative energy levels of donor and acceptor material, the energy loss due to the electron transfer can be minimised: For light absorption in the donor, it is hoped that if the energy offset between donor LUMO (lowest unoccupied molecular orbital) and acceptor LUMO is a tiny bit larger than the exciton binding energy, a positive impact on the open-circuit voltage will be seen.
In the figure, the schematic energy level diagram of a bilayer solar cell is shown. The anode is made of TCO (transparent conductive oxide), then follow donor and acceptor, and finally the metal cathode.  The exciton is photogenerated in the donor, which can diffuse to and dissociate at the interface to the acceptor. The resulting polaron pair then is energetically separated by the effective band gap of the organic solar cell, Eg.The smaller the LUMO-LUMO offset & – which still has to be larger than the exciton binding energy – the larger Eg: the open circuit voltage is maximised, as it equals Eg minus band bending BB and the injection barriers phi [Cheyns 2008].
The exciton is photogenerated in the donor, which can diffuse to and dissociate at the interface to the acceptor. The resulting polaron pair then is energetically separated by the effective band gap of the organic solar cell, Eg.The smaller the LUMO-LUMO offset & – which still has to be larger than the exciton binding energy – the larger Eg: the open circuit voltage is maximised, as it equals Eg minus band bending BB and the injection barriers phi [Cheyns 2008].
As a side note, recently there was a report that claims that the polaron pair dissociation yield is exponentially proportional to the LUMO-LUMO offset [Ohkita 2008]. This implies that always a trade-off between short circuit current and open circuit voltage will have to be made. Please note, however, that the photoinduced absorption measurements by Shuttle et al. are done without the application of an external (or internal for that matter, the samples being optical thin films without electrodes) electric field; so there is still hope;-)
But back to absorption and its impact on the power conversion efficiency: you may know the detailed balance calculation of Shockley and Queisser for inorganic solar cells, power conversion efficiency vs band gap, which has been mentioned recently. 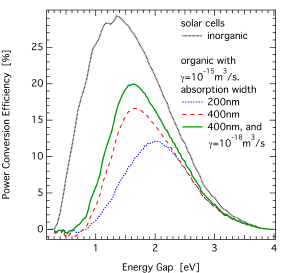 It is shown in the left figure as black solid line. For organic solar cells, there is no complete analytic theory to describe all parameters proportional to the properties, therefore I made an estimate based on a couple of assumptions:
It is shown in the left figure as black solid line. For organic solar cells, there is no complete analytic theory to describe all parameters proportional to the properties, therefore I made an estimate based on a couple of assumptions:
- quantum efficiency 100% within absorption band of 200 (blue) or 400nm (red) width
- fill factor 80%
- thickness 200nm
- open circuit voltage after [Koster 2005] with recombination strength gamma either 10-15m3/s (as in the article, even if not explicitly mentioned) or a reduced recombination strength of 10-18m3/s (green); the other parameters are as in the paper.
- Koster’s Eg is set to “Energy Gap” (as in “x axis”) minus exciton binding energy Eb of 0.3eV
Clearly, disordered organic solar cells have a lot of potential in view of manufacturing by roll-to-roll printing, but the maximum power conversion efficiencies which can be reached are lower as compared to inorganic solar cells… unless a clever chemist (actually, we need a genius here;-) manages to make suitable donor and acceptor materials with wider absorption ranges… Of course, higher intrinsic absorption is not the only route for higher organic photovoltaic performances: another possibility are (solution-processed!) multijunction solar cells, thus combining different absorption ranges of already existing materials.
Update 24.3.2008: I exchanged the wrong reference Shuttle 2008 by Ohkita 2008. Apologies!

For me another big trade-off is wanting a lower bandgap material to increase light absorption while still having a material which self organises into a (semi) crystalline state which obviously helps with charge transport. For me, a donor-acceptor type polymer will give you a lower bandgap (e.g. < 1 eV) but I doubt if such a polymer will ever be strongly crystalline. On the other hand I think I read somewhere that a more homogeneous polymer which could have strong crystallising ability will never have a band-gap below 1.5 eV. I forget the reference but I have it somewhere on my desk. For me again the choice between the two is clear. Charge transport at this time is much more critical than light absorption and so we need to develop a homopolymer with a bandgap as close to 1.5 eV as possible (and of course has good charge transport).