In order to improve the power conversion efficiency of organic solar cells, novel donor and acceptor materials will have to be synthesised. 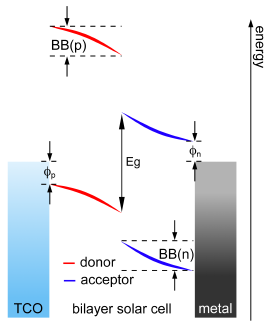 Properties looked for are the ability to self-organise – enhancing order and thus charge transport – and an absorption spectrum as wide as possible, being one of the major limiting factors as of yet. Nowadays, in most cases only the donor material absorbs light efficiently; an absorbing acceptor has a large potential for increasing the photocurrent. Additionally, by a variation of the relative energy levels of donor and acceptor material, the energy loss due to the electron transfer can be minimised: For light absorption in the donor, it is hoped that if the energy offset between donor LUMO (lowest unoccupied molecular orbital) and acceptor LUMO is a tiny bit larger than the exciton binding energy, a positive impact on the open-circuit voltage will be seen.
Properties looked for are the ability to self-organise – enhancing order and thus charge transport – and an absorption spectrum as wide as possible, being one of the major limiting factors as of yet. Nowadays, in most cases only the donor material absorbs light efficiently; an absorbing acceptor has a large potential for increasing the photocurrent. Additionally, by a variation of the relative energy levels of donor and acceptor material, the energy loss due to the electron transfer can be minimised: For light absorption in the donor, it is hoped that if the energy offset between donor LUMO (lowest unoccupied molecular orbital) and acceptor LUMO is a tiny bit larger than the exciton binding energy, a positive impact on the open-circuit voltage will be seen.
In the figure, the schematic energy level diagram of a bilayer solar cell is shown. The anode is made of TCO (transparent conductive oxide), then follow donor and acceptor, and finally the metal cathode.  The exciton is photogenerated in the donor, which can diffuse to and dissociate at the interface to the acceptor. The resulting polaron pair then is energetically separated by the effective band gap of the organic solar cell, Eg.The smaller the LUMO-LUMO offset & – which still has to be larger than the exciton binding energy – the larger Eg: the open circuit voltage is maximised, as it equals Eg minus band bending BB and the injection barriers phi [Cheyns 2008].
The exciton is photogenerated in the donor, which can diffuse to and dissociate at the interface to the acceptor. The resulting polaron pair then is energetically separated by the effective band gap of the organic solar cell, Eg.The smaller the LUMO-LUMO offset & – which still has to be larger than the exciton binding energy – the larger Eg: the open circuit voltage is maximised, as it equals Eg minus band bending BB and the injection barriers phi [Cheyns 2008].
Continue reading “Optimisation Routes for Organic Solar Cells – Absorption”


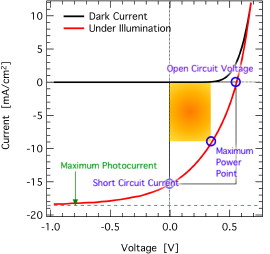 made for crystalline inorganic diodes, when applied on fitting organic solar cells.
made for crystalline inorganic diodes, when applied on fitting organic solar cells.

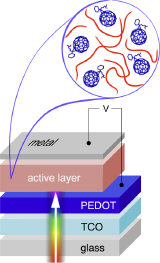 The vision for the so called plastic electronics is to print circuits and devices on flexible substrates. This can be done at room temperature (low energy) and ideally with roll-to-roll processes (high throughput). Sounds good, eh?
The vision for the so called plastic electronics is to print circuits and devices on flexible substrates. This can be done at room temperature (low energy) and ideally with roll-to-roll processes (high throughput). Sounds good, eh?